Classic clinical trials are usually insufficient to support the kind of claims associated with stem cell activity. Margret Ebauer introduces novel in vitro tools for evaluating the effects of such cosmetic actives
Efficacy data for skin care actives is one of the must-haves to sell a product. However, the added value to R&D and marketing greatly depends on accurate models and scientifically proven data. Clinical evaluation studies are the best, but for many new claims, eg stem cell protection, they are hardly applicable. Here, in vitro data is a useful indicator and this article will illustrate the latest trends in and concepts for in vitro testing and their use for stem cell related claims.
What are stem cells?
Adult stem cells are located in specific niches within each tissue. They may be thought of as a specialised sub-population of progenitor cells that show the unique ability to self-renew indefinitely and are thus the only cells to be active throughout our entire life. In the skin, they can be found both in the epidermis (in the bulge region of the hair follicle and in the basal layer) and in the dermis (eg around the dermal papilla, figure 1).
The different compartments of the skin are continually renewed as cells age in a continuous process driven by the resident stem cell population. Epidermal stem cells are now known to regenerate the epidermis, hair follicles and sebaceous glands. Dermal stem cells play an equally important anti-ageing role by regenerating the stromal cells and thus collagen matrix of the dermis. In this way both stem cell types found in the skin play important anti-ageing and regenerative roles.[1,2]
Stem cell related claims
Significant advances have been made in our understanding of the mechanisms underlying skin renewal and regeneration. Numerous studies dealing with regulation and signalling pathways involved in stem cell differentiation, regulation and self-renewal have appeared to date. As a result, anti-ageing cosmeceuticals are increasingly targeting the protection and reactivation of epidermal stem and progenitor cells in the skin. Other approaches such as improvement of migration capacity or optimisation of regeneration capacity have also been identified as powerful mechanisms to delay the ageing processes.
Dermal stem and progenitor cells have also been targeted by cosmeceuticals as they play an important role in the balance and regeneration of the dermis. The maintenance of a sufficient number of dermal progenitor cells, prevention of their ageing or restoration of their activity would be expected to lead to a reinforcement of the extracellular matrix, thus combating one of the most visible signs of ageing. In view of the way epidermal and dermal stem cells collaborate to maintain skin integrity, approaches are now beginning to emerge that could make global rejuvenation of the skin possible.[3]
As a consequence, a range of active ingredients – including stem cell stimulating peptides, amino acids and enzymes ranging from growth factors to telomerase – have been developed for use in topical formulations. A wide range of plant-derived natural extracts or biotechnologically produced plant stem cells which claim to protect epidermal stem cells have also been recently developed. However, specific approaches are required to support such specialised claims.
Stem and progenitor cells represent only a small fraction of the total cells in the epidermis, making it very difficult to demonstrate an effect on these cells in vivo. In addition, to demonstrate mechanistic effects in human tissue requires the use of tissue samples or biopsies, which are often expensive, irregularly available and incompatible with certain analytical techniques.
Thus classic methods, ie clinical trials, are typically insufficient to support novel claims of stem cell associated activity. Novel in vitro tools are needed for evaluation of the effect of cosmetic actives directly on the progenitor and stem cells involved in skin renewal.
Novel substantiation tools
Using a new technique known as Progenitor Cell Targeting (PCT), it is now possible to establish enriched cultures of progenitor cells and maintain them for extended periods in vitro. This technique uses specialised culture media designed to select particular cell populations, such as progenitor cells, by accurately mimicking the microenvironment these cells experience in vivo, known as the niche environment.[4]
Cells established using this technique can be used to test the effect of an extract in either 2D or 3D culture. Mechanistic evaluations can be made using a full range of molecular and cell biology techniques, and in addition more stem cell-specific evaluations can be performed, such as colony forming efficiency (CFE) in 2D culture. CFE is an excellent indicator of progenitor cell function as only these undifferentiated cell types retain the ability to form colonies in vitro. For these tests cells are seeded at low density and then allowed to form colonies. Colonies are then counted and the percentage of cells seeded that formed colonies calculated. This method is effectively able to demonstrate an extract’s ability to maintain stem cell potential even after an environmental challenge or insult, like UV irradiation or oxidative stress.
A
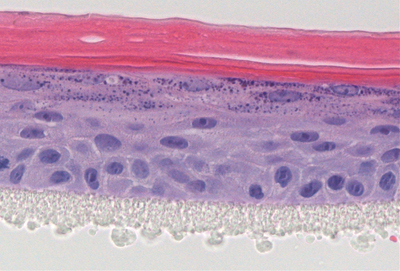
Figure 2: Cross section of a 3D epidermal model, established using primary human keratinocytes exposed to UV-induced stress. Topical treatment with a test compound (A) protected cells from UV stress, normal 3D epidermal structure was formed. Above the plastic membrane (bottom layer) the complete epidermal structure is visible, including the basal, spinous, granular and cornified layers. Unprotected control (B) showed incomplete, disrupted morphology
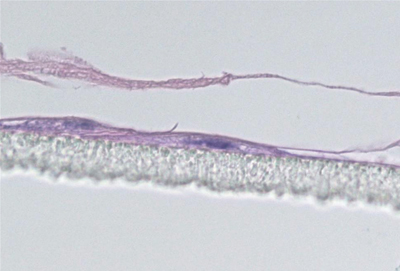
Maintenance of the progenitor cell’s ability to form 3D structures is another target oriented approach as only progenitor cells retain the ability to form complex 3D structures in vitro. In this approach, cells are first exposed to an environmental stress. Subsequently cells are seeded onto a permeable membrane and then cultured at the air-liquid interface to establish a 3D culture. The protective effect of the extract treatment on progenitor cells is then easily seen in the generation of a normal epidermal structure (figure 2).
Progenitor cells derived from the dermis have been found to exhibit various stem cell-like characteristics, for example generation of new dermal cells for tissue maintenance, wound healing and hair follicle morphogenesis. They are also able to form spheres.[5]
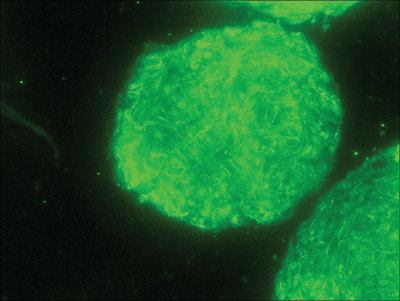
Recent research has shown that these cells reside in the dermal papillae of the hair follicle and express specific markers like Sox2 and versican (figure 3).
A
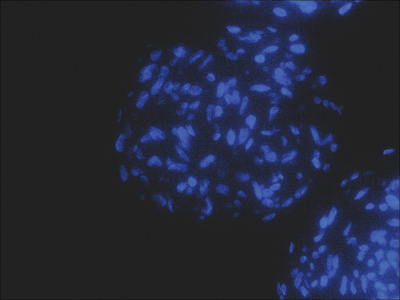
Figure 3: (A) Dermal stem cell marker Sox2 (green) detection in dermal papilla derived cells. (B) Nuclear staining (blue) was used as counter stain
New high performance products with powerful claims need to prove their activity via scientific research and novel efficacy studies. The combination of in vivo and in vitro approaches can demonstrate not only visible effects of a compound but also its influence on underlying regenerative mechanisms.
Author
Margret Ebauer
Manager contract research & testing
CELLnTEC Advanced Cell Systems AG
Switzerland
tel: +41 31 331 9582
email margret@cellntec.com
www.cellntec.com
References
1. Lev V, Lindon C, Zheng Y, Harfe BD & Morgan BA, Epidermal stem cells arise from the hair follicle after wounding, FASEB J (21) 1358-1366 (2007)
2. Nowak JA, Polak l, Pasolli HA & Fuchs E, Hair Follicle Stem Cells Are Specified and Function in Early Skin Morphogenesis, Cell Stem Cell (3) 33-43 (2008)
3. Zouboulis CC, Adjaye J, Akamatsu H, Hoe-Behrens G & Niemann C, Human skin stem cells and the ageing process. Experimental Gerontology, (43) 986-9 (2008)
4. Lemischka IR & Moore KA, Stem cells: interactive niches, (425) 778-779 (2003)
5. Biernaskie J, Paris M, Morozova O, Fagan BM, Marra M, Pevny L & Miller F, SKPs Derive from Hair Follicle Precursors and Exhibit Properties of Adult Dermal Stem Cells, Cell Stem Cell, 5, 610-623 (2009)



